Obligations of EU Importers
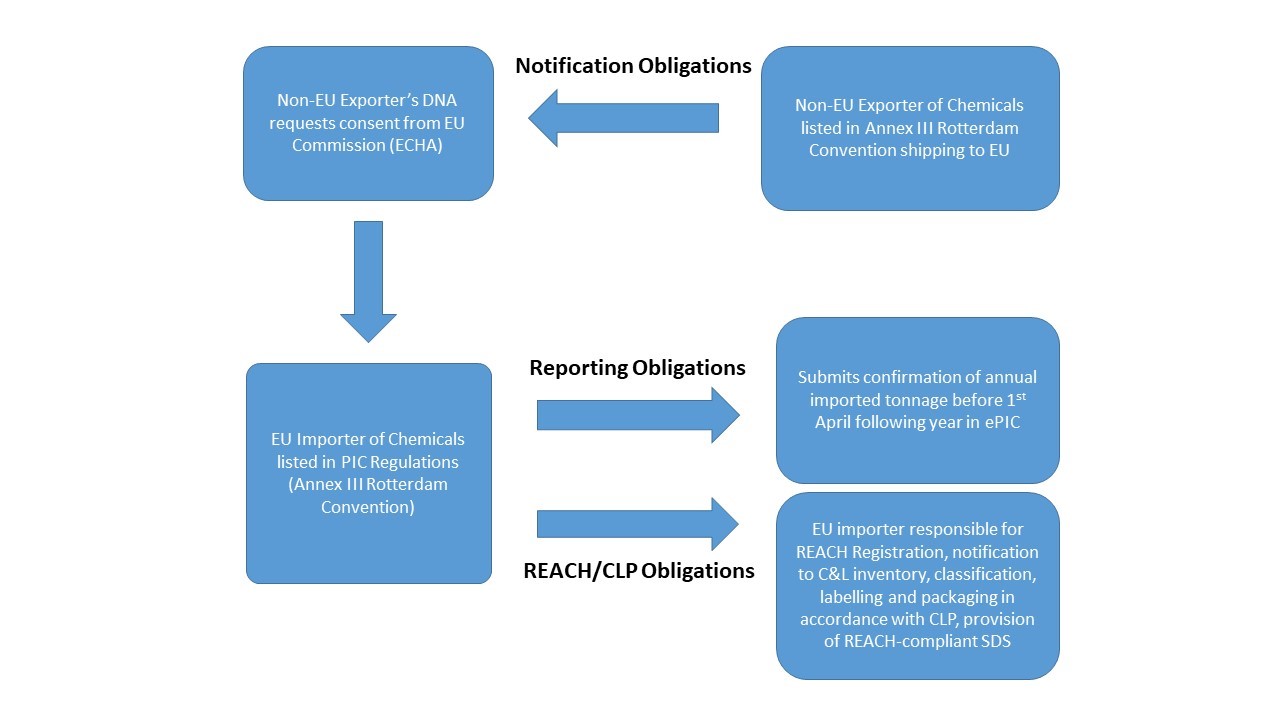
Notification Obligations
The non-EU exporter of chemicals listed in Annex III to the Rotterdam Convention (also listed in Annex I part 3 of the Export Import Regulations) is required to submit an export notification to the relevant DNA in their country. The DNA then requests ECHA, on behalf of the EU Commission, to provide consent. ECHA provides a response to the DNA, and notifies the DNA of the relevant EU Member State of the import.
Reporting Obligations
By 30th March each year, all EU importers of chemicals listed in Annex I of the Export Import Regulation shall inform their DNA of the actual quantity of:
(a) substances listed in Annex I;
(b) mixtures containing such substances in a concentration that triggers labelling obligations under CLP irrespective of the presence of any other substances; or
(c) articles containing substances listed in Part 2 or 3 of Annex I in unreacted form or mixtures containing such substances in a concentration that triggers labelling obligations under CLP irrespective of the presence of any other substances;
Exported or imported in the previous calendar year using ePIC (Article 10 report) regardless of the intended use of the chemical. The ePIC Industry User Manual provides advice on completing this report.
REACH/CLP Obligations
For importers, the primary legislations for industrial chemicals are the REACH and CLP Regulations. For an overview of how importers can comply, please see our information sheet.
Further Information
To assist you in determining your role under our chemical legislation, please visit our 'Role Finder Tool'.
